Medical Waste Disposal Laws
Since 1999, Med Monkey has been a fully licensed transporter of biohazardous waste, making us the perfect option for all of your medical waste disposal needs in central Indiana. We are proud of our commitment to ethical business practices, as well as our devotion to exceptional customer care. Contact us today for a free quote of our medical waste disposal services by calling (317) 855-9248.
Our Commitment to State & Federal Waste Regulations
Med Monkey is ready and available to support you in the removal of your medical waste. As a permitted transporter of biohazardous waste, we are required by law to comply with all state and federal waste regulation laws.
Whether your facility is a hospital, medical clinic, dentist office, nursing home, funeral home, laboratory, veterinary clinic, body-piercing salon, tattoo shop, or any other biomedical waste generator, your organization is legally bound by law to follow the same state and federal waste disposal regulations as well.
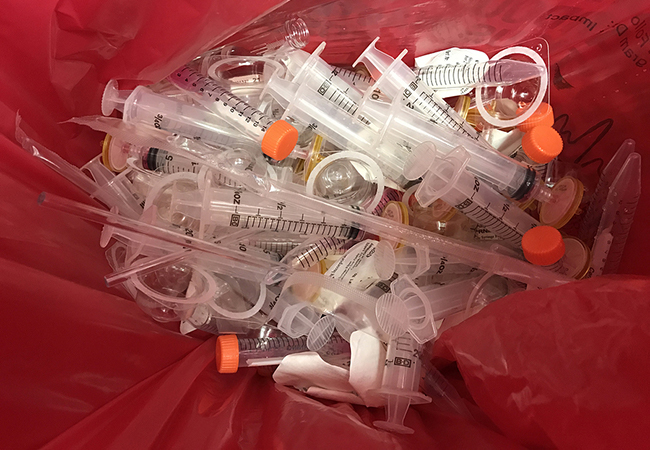
Med Monkey’s Waste Acceptance Policy
“Regulated Medical Waste” is a: (1) waste or reusable material derived from medical treatment of an animal or human, which includes diagnosis and immunization, or from biomedical research, which includes production and testing of biological products. Regulated medical waste is assigned to UN 3291. No Category A materials (assigned to UN 2814 or UN 2900 as appropriate) may be discarded in the regulated medical waste containers; (2) As defined in OSHA’s Bloodborne Pathogens Standard – 29 CFR 1910.1030 and all other applicable laws, rules, regulations and guidelines; and (3) trace chemotherapy discarded items which may have been contaminated provided that such items, including vials and syringes, shall be “empty” as defined in applicable laws, regulations, and guidelines.
Quick Reference Acceptable or Non-Acceptable Regulated Medical Waste
The Following Waste and/or Materials WILL NOT BE ACCEPTED by Med Monkey:
- Items that don’t fall under the definition of Regulated Medical Waste
- Sharps not properly contained in sharps containers
- Listed or characteristically hazardous wastes
- Hazardous chemotherapy waste
- Radioactive waste
- Complete human remains
- Any item or material containing Mercury
- Any item or material containing Amalgam
- Any items containing batteries including Pacemakers and Bovie cauterizing pens
- Any flammable liquids or empty containers with flammable liquids or aerosol cans
- Any corrosive or caustic materials
Customer Packaging: Customer represents and warrants to the Transporter to package all Regulated, Infectious, Chemotherapeutic, and Pathological waste in accordance to all Local, State, and Federal regulations. The customer further agrees not to place any Hazardous, Radioactive, Toxic, Explosive, Corrosive materials to be picked up by Med Monkey. All containers must be lined with a red bag. Before closing the container, the red bag must be tied off in a single knot in order to contain the waste inside.
Unacceptable Material for Regulated Medical Waste.
- Any material not considered Regulated/Infectious Medical Waste or Sharps as defined above.
- Any material possessing the characteristics of, or containing materials regulated for, flammability, corrosiveness, reactivity or EP toxicity as defined by the Environmental Protection Agency Regulations for Defining Hazardous Waste in CFR 261.21 through 261.24.
- Any material or substance considered hazardous or toxic as defined by applicable federal, state, or provincial laws or regulations.
- Any compressed gas cylinders or aerosol containers.
- Any materials regulated by the Nuclear Regulatory Commission and/or the Department of Transportation as radioactive. However, it should be noted that materials containing the isotopes tritium (H3) or Carbon 14 (C14) having an average value below 0.002 micro curie/gram are not regulated by the NRC, DOT or most state agencies and therefore may be acceptable to place in MedWaste containers.
- Human torso including fetal torso.
- Unacceptable waste will also include medical waste that is inappropriately packaged.
Customer shall not place the following items inside any waste container provided by Med Monkey: Customer acknowledges that Med Monkey does not have the ability to treat the following: (a) bulk pharmaceutical waste; (b) RCRA characteristic chemotherapy waste which has an alcohol base that makes the mixture ignitable; (c) Chlorambucil, Cyclophosphamide, Daunomycin, Melphalan, Mitomycin C, Streptozotocin, and Uracil Mustard (which are listed RCRA Hazardous Wastes and must be managed as such); (d) human fetuses
All Sharp objects must be placed in a Sharps container with a lid. The sharps container will be placed in a Red bag to be transported in a DOT-approved container.
Internet Resources
-
Veterinary Guidelines:
- Veterinary Compliance Assistance
- Cornell University Bio Waste Disposal Guide
Funeral Home Waste Stream
Pacemakers cannot be processed. Pacemakers are to be sent back to the manufacturer. Alternatively, pacemakers can be donated to organizations such as Pace 4 Life or My Heart Your Heart .
Funeral homes utilize various hazardous chemicals for embalming purposes. Many of these chemicals are flammable, toxic, or acidic. In short, these chemicals are considered acutely hazardous since they have the designated hazardous code H. In order for the used containers to be considered RCRA empty, the EPA requires the containers to be triple rinsed ((§261.7(b)(3)(i)). When the containers are triple rinsed, please have the generators leave the bottles uncapped then placed in the lined biohazardous container.
Med Monkey gladly serves Richmond, Westfield, and the surrounding areas of Indiana. Call our office at


